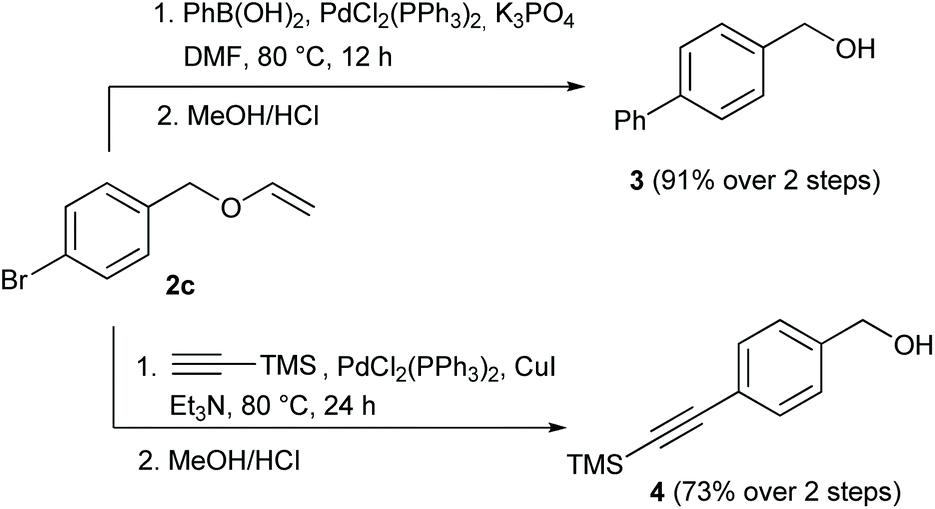
Examining the vinyl moiety as a protecting group for hydroxyl (–OH) functionality under basic conditions - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D0QO00202J

Examining the vinyl moiety as a protecting group for hydroxyl (–OH) functionality under basic conditions - Organic Chemistry Frontiers (RSC Publishing)

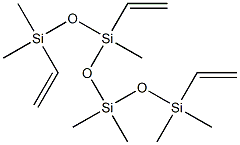
Formation of Linear Polymers with Pendant Vinyl Groups via Inclusion Complex Mediated Polymerization of Divinyl Monomers | Journal of the American Chemical Society
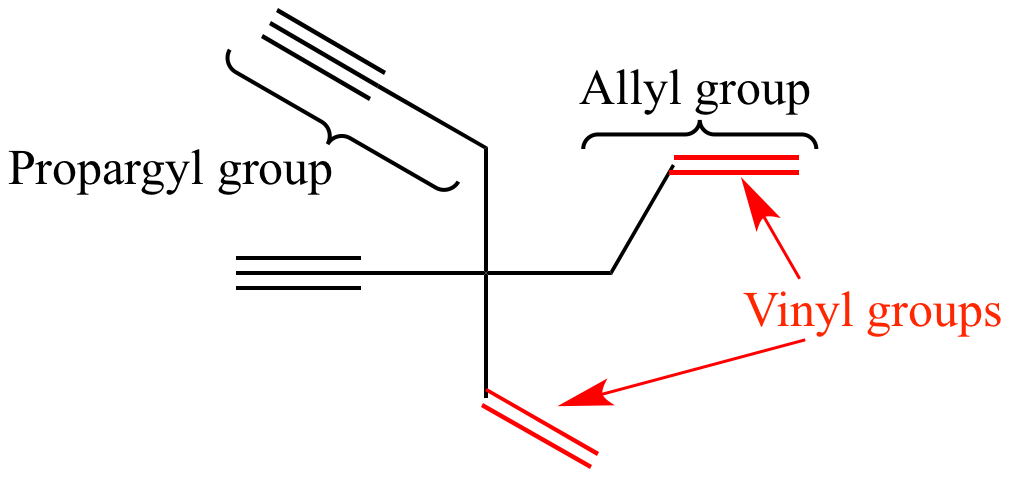
 \\[{\\text{HC}} \\equiv {\\text{C}} - \\](C) \\[{{\\text{H}}_2}{\\text{C}} = {\\text{CH}} - {\\text{C}}{{\\text{H}}_2} - \\](D) \\[{\\text ... Which of the following is a vinyl group?(A) \\[\\left( {{\\text{C}}{{\\text{H}}_3}} \\right){\\text{CH}} - \\](B) \\[{\\text{HC}} \\equiv {\\text{C}} - \\](C) \\[{{\\text{H}}_2}{\\text{C}} = {\\text{CH}} - {\\text{C}}{{\\text{H}}_2} - \\](D) \\[{\\text ...](https://www.vedantu.com/question-sets/2353ddd8-d29e-491b-8fd2-4bc801a90c5b2135321051813214169.png)
Which of the following is a vinyl group?(A) \\[\\left( {{\\text{C}}{{\\text{H}}_3}} \\right){\\text{CH}} - \\](B) \\[{\\text{HC}} \\equiv {\\text{C}} - \\](C) \\[{{\\text{H}}_2}{\\text{C}} = {\\text{CH}} - {\\text{C}}{{\\text{H}}_2} - \\](D) \\[{\\text ...
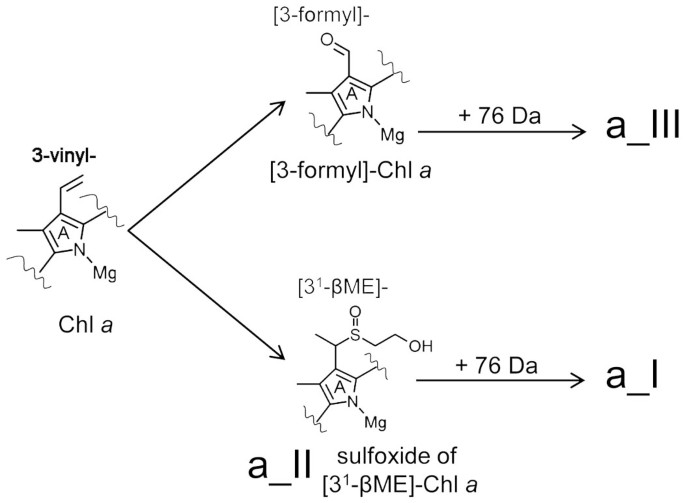
In vitro Conversion of Vinyl to Formyl Groups in Naturally Occurring Chlorophylls | Scientific Reports

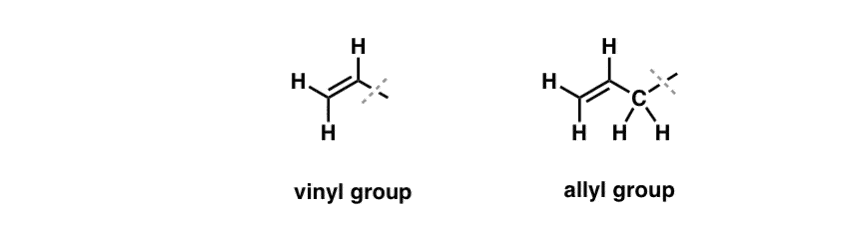
Vinyl group Allyl group Functional group Organic chemistry Ethylene, Over Fifty Group, angle, text, rectangle png | PNGWing



:max_bytes(150000):strip_icc()/vinylgroup-58b5e08d5f9b586046f45384.jpg)
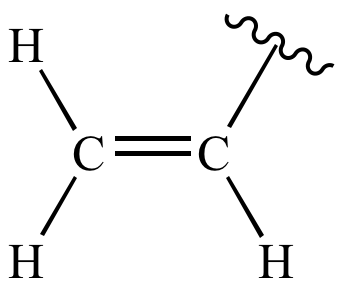






![유기화합물 명명법] 비닐기. vinyl group 유기화합물 명명법] 비닐기. vinyl group](https://t1.daumcdn.net/cfile/tistory/99850D395EBF4B1C31)


